 Chemists
have a special shorthand way of writing the names of chemicals. For example,
water has the formula H2O and salt is NaCl. When iron combines
with oxygen in the air to rust, chemists write an equation:
Chemists
have a special shorthand way of writing the names of chemicals. For example,
water has the formula H2O and salt is NaCl. When iron combines
with oxygen in the air to rust, chemists write an equation:
4 Fe + 3
O2 ![]() 2 Fe2O3. Like any shorthand
method of writing, with a few simple rules, the code can be broken and
easily read.
2 Fe2O3. Like any shorthand
method of writing, with a few simple rules, the code can be broken and
easily read.
This topic will take you through the rules of writing formulas and will help you to read what a chemist means when they write formulas. The next topic Reading chemical equations will help you to read and understand chemical equations.
Formulas
Chemists have formulas or shorthand names for all atoms, ions, molecules
and ionic salts known. If a new substance is discovered, a new formulas
is used to describe it using the code of formulas writing.
Rule 1:- All substances are made of atoms. There are 92 naturally occurring atoms, each with their own formulas. For example, the simplest atom, hydrogen is written simply as H, while the next atom, helium, is He. A full list of the elements and their formulas can be found in most science texts under the title "Periodic Table of the elements". See the related web sites for more on the elements and their formulas.
All elements are given a symbol that is either a single capital letter or a capital followed by one or two lower case letters. Here is a list of a few of the elements and their formulas that will be used in this topic:
|
hydrogen
|
H
|
sulfur
|
S
|
|
helium
|
He
|
silicon
|
Si
|
|
carbon
|
C
|
sodium
|
Na
|
|
calcium
|
Ca
|
aluminium
|
Al
|
|
chlorine
|
Cl
|
oxygen
|
O
|
|
copper
|
Cu
|
iron
|
Fe
|
Most formulas reflect the English name of the element, but some follow either the Latin or an older name. For example, Cu for copper comes from its Latin name 'cuprum' and Fe for iron comes from the Latin 'ferrum'.
Rule 2: Formulae of molecules.
When writing the formula for a molecule, like H2O for water,
all the atoms present in the molecule are listed. A subscript number follows
any atoms which are present in the molecule more than once. Formulae are
written with no gaps and the capitals denote a new element. 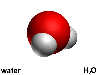
In the water molecule there are two hydrogen atoms and one oxygen atom.

Copper sulfate has the formula:- CuSO4 which represents 1 copper
atom, 1 sulfur atom and 4 oxygen atoms.
Here are some more:-
|
sodium chloride
|
salt
|
NaCl
|
1 sodium and 1 chlorine atom
|
|
silicon dioxide
|
sand
|
SiO2
|
1 silicon and 2 oxygen atoms
|
|
aluminium oxide
|
alumina
|
Al2O3
|
2 aluminium and 3 oxygen atoms
|
Rule 3:- Formula of ions. Ions are electrically charged atoms or molecules. They are formed when electrons are either added or removed from an atom or molecule. The charges on ion are shown with a superscript number, if more than one, and a sign. For example, the sodium ion has a charge of 1+, hence its formula is Na+. Oxygen forms a 2- ion, its formula is O2-.
Here are a few more ions and their formulas:
|
hydrogen ion 1+
|
H+
|
chloride ion 1-
|
Cl-
|
|
calcium ion 2+
|
Ca2+
|
sulfide ion 2-
|
S2-
|
|
aluminium ion 3+
|
Al3+
|
sulfate ion 2-
|
SO42-
|
Note: The sulfate ion's formula (SO42-) shows it has 1 sulfur atom and 4 oxygen atoms with an overall charge of 2-.
Rule 4:- Formulae of salts. When ions combine to form salts, the superscript charges are not shown and only the atoms and their rations are displayed, just like for molecules. Sodium chloride and copper sulfate mentioned above are both ionic salts.
Now go to next topic Reading chemical equations which will help you to read and understand chemical equations.
| Copyright owned by the State of Victoria (Department of Education and Early Childhood Development). Used with Permission. |