|
The force of steam
When your kettle boils, water evaporates and becomes a gas called
water vapour - the steam you see coming from the spout is this vapour
condensing again in the cooler air. Water vapour has a property that
is so very important in everyday life; it takes up much more volume
than the liquid water it came from. The force produced by steam is so
powerful that it can drive large steam engines and turn turbines of
power stations to generate electricity.
The steam engine
 An Egyptian named Hero developed a steam engine about 120 BC. He
made a small hollow globe with a pipe through the centre connected to
a vessel producing steam. On both sides of the sphere he fixed two L-shaped
tubes so that the steam issued from them and caused the sphere to spin.
It was not able to do any real work but did demonstrate the power of
steam.
An Egyptian named Hero developed a steam engine about 120 BC. He
made a small hollow globe with a pipe through the centre connected to
a vessel producing steam. On both sides of the sphere he fixed two L-shaped
tubes so that the steam issued from them and caused the sphere to spin.
It was not able to do any real work but did demonstrate the power of
steam.
 James
Watt was said to have thought of the idea of a steam engine by watching
steam issuing from a kettle of boiling water - not likely but a good
story! James
Watt was said to have thought of the idea of a steam engine by watching
steam issuing from a kettle of boiling water - not likely but a good
story!
 James Watt actually improved on existing steam engines, taking
out his first patent in 1769. Further improvements by many engineers
over the years culminated in the steam engines that could power locomotives
and the propellers of ocean liners and freighters.
James Watt actually improved on existing steam engines, taking
out his first patent in 1769. Further improvements by many engineers
over the years culminated in the steam engines that could power locomotives
and the propellers of ocean liners and freighters.
Thermal power stations
Nuclear, coal powered, oil and gas burning power stations all work by
heating water under pressure to create high energy steam. This high
pressure, high temperature steam is used to turn turbines which, when
attached to electrical generators, produce useful electrical energy.
The vital link in the transformation of the stored potential energy
in the fuel and the electrical energy produced is the ability of steam
to efficiently transfer thermal energy.
Cooking under pressure
 In a pressure cooker, water is heated and a weight is placed on
the only exit from an otherwise sealed unit. Steam produced in the pressure
cooker forces against the weight and the pressure inside the cooker
increases until the weight is lifted. Depending on the weight used,
pressures much higher than the normal 1 atmosphere pressure can be reached
inside the cooker. Water boils when the vapour pressure exceeds the
atmospheric pressure. By increasing the pressure within the cooker,
the energy needed by the water molecules
to boil has to increase as well. Hence, the
water boils at a higher temperature. The food
gets much hotter by this process and therefore it cooks faster.
In a pressure cooker, water is heated and a weight is placed on
the only exit from an otherwise sealed unit. Steam produced in the pressure
cooker forces against the weight and the pressure inside the cooker
increases until the weight is lifted. Depending on the weight used,
pressures much higher than the normal 1 atmosphere pressure can be reached
inside the cooker. Water boils when the vapour pressure exceeds the
atmospheric pressure. By increasing the pressure within the cooker,
the energy needed by the water molecules
to boil has to increase as well. Hence, the
water boils at a higher temperature. The food
gets much hotter by this process and therefore it cooks faster. 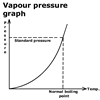
Vapour pressure and boiling
point
There is a simple relationship between vapour pressure and boiling point.
This graph shows how the boiling point varies with vapour pressure.
As the vapour pressure inside the cooker is increased, the temperature
at which the liquid boils also increases.
|