|
The hole in the ozone layer B: The
players
"The hole in the ozone layer" sounds like a title of a "B
Grade" horror movie, conjuring up all 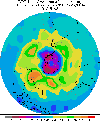 sorts
of misconceptions. It is not a hole in the atmosphere, but a depletion
of ozone above the polar regions which occurs in the spring and summer
months. sorts
of misconceptions. It is not a hole in the atmosphere, but a depletion
of ozone above the polar regions which occurs in the spring and summer
months.
Not to be confused with "The greenhouse effect", ozone depletion
seems to be a relatively new phenomenon, with scientists first suspecting
there was a problem in the late 1970s. It was not until 1985 that British
scientists discovered the first "hole" over Antarctica.
The hole
in the ozone layer A: What is it? investigates how the "hole"
forms and what is being done to halt its growth. The second part of
this topic will look at the players in the formation of the "hole";
ozone, chlorofluorocarbons and ultraviolet light. T
Ozone
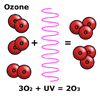 Ozone,
O3, is a form of oxygen gas, which is produced when ultraviolet
light from the Sun strikes and splits an oxygen molecule, O2,
to form two oxygen atoms. These atoms then combine with other oxygen
molecules to form ozone. Ozone,
O3, is a form of oxygen gas, which is produced when ultraviolet
light from the Sun strikes and splits an oxygen molecule, O2,
to form two oxygen atoms. These atoms then combine with other oxygen
molecules to form ozone.
The high intensity of ultraviolet radiation (UV) from the Sun striking
the upper atmosphere ensures that ozone is readily generated in the
Earth's upper atmosphere. Unfortunately, ozone is not very stable and
can be destroyed by more ultraviolet radiation or contact with other
chemicals, especially chlorine atoms.
Ozone can exist fleetingly in the lower atmosphere, soon reacting with
other chemicals. Ozone is also created by lightning and electrical discharges
and is the odour that can be smelt near arc welding and older photocopying
machines.
CFCs: the ozone destroyers
 The
destruction of the ozone in the upper atmosphere is being caused by
a group of chemicals called chlorofluorocarbons or CFCs. CFCs are very
stable, unreactive gases once used as aerosol propellent gases, refrigerants
and in the electronic industry. These gases were used because they were
so unreactive and safe to use in the home. CFCs are still in use in
small quantities, but their use is slowly being phased out. The
destruction of the ozone in the upper atmosphere is being caused by
a group of chemicals called chlorofluorocarbons or CFCs. CFCs are very
stable, unreactive gases once used as aerosol propellent gases, refrigerants
and in the electronic industry. These gases were used because they were
so unreactive and safe to use in the home. CFCs are still in use in
small quantities, but their use is slowly being phased out.
Because CFCs do not degrade and break down in the atmosphere, they
simply accumulated until they reach the upper atmosphere (this can take
years and even decades for some of the heavier CFCs). At this point
they turn from "Dr Jekyll" to "Mr Hyde". CFCs break
apart when struck by ultraviolet light forming different bits of molecules
and chlorine atoms. It is these free chlorine atoms, known as radicals,
which are so dangerous to the ozone layer.
 Ultraviolet light
Ultraviolet light
The third constituent in the ozone drama is ultraviolet light or UV.
This high-energy electromagnetic radiation from the Sun has the ability
to break molecules apart. It is UV that causes sunburn, skin cancer
and blindness from cataracts on the eye. Astronauts must wear protective
space suits to be able survive the high intensity UV coming from our
Sun in outer space. The ozone layer is the Earth's "space suit"
and without it life on Earth would not exist in its present form.
|