|
States of matter
All matter (anything made up of atoms) is divided into four physical
"states": solid, liquid, gas and plasma. The chemical composition
of a substance determines its state at any particular temperature. 
All substances start as solids at the very lowest possible temperature,
absolute zero
(-273°C) and go through changes of state as they are
heated. As a solid is heated, it will generally melt to become a liquid,
boil to form a gas and finally, at extreme temperatures like those found
on the Sun or a very hot flame, its atoms will break apart to become
a plasma.
The states of matter on Earth
 Plasma, in the form of ionised gas, is very rare
on Earth and usually only exists fleetingly as the glowing gas in a
flame or as a bolt of lightning. Gases are mainly found in the Earth's
atmosphere, although small pockets of gas are also caught in the Earth's
mantle. Most of the outer part of our planet is solid; this solid mantle
sits on an outer liquid core and a solid inner core. Two thirds of the
Earth's surface is covered in liquid water.
Plasma, in the form of ionised gas, is very rare
on Earth and usually only exists fleetingly as the glowing gas in a
flame or as a bolt of lightning. Gases are mainly found in the Earth's
atmosphere, although small pockets of gas are also caught in the Earth's
mantle. Most of the outer part of our planet is solid; this solid mantle
sits on an outer liquid core and a solid inner core. Two thirds of the
Earth's surface is covered in liquid water.
The states of matter in the Universe
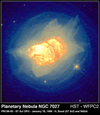 The most common state in the Universe is plasma. This is because
most of the atomic matter in the Universe is found in stars and stars
are massive, extremely hot balls of ionised gas or plasma.
The most common state in the Universe is plasma. This is because
most of the atomic matter in the Universe is found in stars and stars
are massive, extremely hot balls of ionised gas or plasma.
Gas is the next most universally common state. It is found in vast
gas nebula clouds spread throughout space and in the giant gas planets
like Jupiter and Saturn.
Solid forms the third most common state. It is found in the form of
cosmic dust, Earth-like planets, comets and other stray pieces of rock.
Liquid is probably the rarest state in the Universe, with the only
discovered naturally occurring liquids being the Earth's surface water
and our liquid metal core. Some astronomists believe that there may
be water on a few of the moons in the outer Solar System and that water
may have once existed on Mars and carved out its extensive canyons.
What makes one state different from
another?
The state of a substance at any particular temperature and pressure
is dependent on how big its particles are and how strongly they bond
together. For example, substances which are solids under normal conditions
(25°C and 1 atmosphere pressure) are usually made of large heavy
atoms or molecules and are tightly bonded together. On the other hand,
gases under these same conditions tend to be made of small, light atoms
or very small molecules that do not stick together well. Liquids generally
have particles which are somewhere in between these two extremes.
Changing state does not change the
substance
 When
substances undergo a change of state they generally do not chemically
change, or form new substances. They only undergo physical changes.
Water is made of H2O molecules, whether it is ice, liquid
water or steam. Changes in state are usually easily reversed, by either
heating or cooling, whereas chemical changes are more difficult to reverse. When
substances undergo a change of state they generally do not chemically
change, or form new substances. They only undergo physical changes.
Water is made of H2O molecules, whether it is ice, liquid
water or steam. Changes in state are usually easily reversed, by either
heating or cooling, whereas chemical changes are more difficult to reverse.
|