|
Soap and detergents
This topic explores the fascinating world of soaps and how they clean
dirt from clothes and food from dishes. This topic should be read in
conjunction with the topics Like
dissolves like and Talking
solutions.
What is dirt?
Most dirt and food stains are composed of oils and
other biological molecules, sometimes mixed with inert materials like
sand. Oils and other biological molecules are hydrocarbons, which chemist
classify as non-polar molecules.
Like other non-polar molecules such as petrol, wax and grease, most
food and dirt is not soluble in water. Soaking greasy plates has little
effect!
Why won't water remove grease and
dirt?
Water is a polar solvent and, as has been discussed in the topic "Like
dissolves like", will mix with, and dissolve, other polar molecules
and ionic salts, but will not dissolve non-polar molecules.
Hence water is not very good at cleaning plates and clothes by itself.
What is required is a way of bridging the gap between the non-polar
oil (which holds the dirt to the fabric / dishes) and the polar water
molecules.
Soap
and detergents - bridging the gap
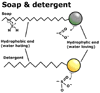 Soap and detergents are quite unique molecules with the ability to dissolve
in both polar and non-polar solvents. Both Soap and detergents are composed
of long non-polar hydrocarbon molecules with a polar or ionic end, thus
they combine both polar and non-polar characteristics in the same molecule.
Soap and detergents are quite unique molecules with the ability to dissolve
in both polar and non-polar solvents. Both Soap and detergents are composed
of long non-polar hydrocarbon molecules with a polar or ionic end, thus
they combine both polar and non-polar characteristics in the same molecule.
This allows the soap and detergent molecules to bond with both oil
and water molecules at the same time, forming a connection between the
oil and the surrounding water molecules. Soap and detergents operate
at the interface between the oil and the water and are known as "surface
active substances".
 With
enough detergent/soap molecules present and agitation, the oil and dirt
will wash off the clothes, attached to the water via the detergent/soap
molecules. The agitation is important as detergent/soap molecules stick
out of the oil like paddles, which catch the "current" and
wash away the oil and dirt with the water. With
enough detergent/soap molecules present and agitation, the oil and dirt
will wash off the clothes, attached to the water via the detergent/soap
molecules. The agitation is important as detergent/soap molecules stick
out of the oil like paddles, which catch the "current" and
wash away the oil and dirt with the water.
Soap and detergents
The basic difference between Soap and detergents is simply that soaps
are solid and detergents are liquid, although the boundary is often
blurred. Soap molecules have longer hydrocarbon chains than detergents
and have different polar ends.
Soap is made by reacting animal fat, stearic acid, with caustic soda,
sodium hydroxide. The chemical name for soap is sodium stearate.
Detergents are made by reacting vegetable oils like lauric acid with
sulfuric acid to produce lauryl sulfate.
Dish washer powders - Warning!
Automatic dishwasher powders are made up of a mixture of soaps and caustic
chemicals like sodium hydroxide. The extra caustic chemicals react with
grease and oil to form more soap molecules, giving a double cleaning
action.
This combination of chemicals makes dish washing powders very efficient,
but also very dangerous. Never touch dishwasher powders and keep them
away from children. They are strong enough to react with oils in skin
and cause damage to eyes. If ingested they will cause severe chemical
burns in the throat and can be fatal.
Dry cleaning
Dry cleaning uses non-water based solvents to clean. Dry cleaning solvents
include, hydrocarbons and organochlorines. These are non-polar molecules
that are capable of dissolving oils from dirty clothes.
|