|
Production of aluminium
Aluminium is probably the most important of the more reactive metals
which requires electrolysis to be extracted from their ore. Its lightness,
strength, resistance to corrosion and good electrical conductivity make
it a very important metal in our modern society.
 Aluminium
forms a protective oxide layer on exposure to air which allows it to
be used in high tension power lines, planes, ships, building and cans
to name but a few. If it were not for this protective oxide layer, its
high reactivity would make it impossible to use safely. Aluminium
forms a protective oxide layer on exposure to air which allows it to
be used in high tension power lines, planes, ships, building and cans
to name but a few. If it were not for this protective oxide layer, its
high reactivity would make it impossible to use safely.
This was shown to be the case in the Falklands War, where many of the
British war ships had aluminium superstructures, because it reduced
their overall weight. When one of these ships was hit with a Cruise
missile, it caught alight and the aluminium started to melt and then
burn itself, totally destroying the ship.
Electrolysis
 The
ore of aluminium is called bauxite, a brown, rusty coloured mineral containing
aluminium oxide or alumina (Al2O3). Bauxite is
usually found in areas of high rainfall and is the product of leaching
aluminium oxide from surface rock over millions of years. Alumina is
first chemically separated from bauxite ore by dissolving the bauxite
in sodium hydroxide solution, filtering off the unwanted coloured components
and then extracting the alumina by precipitation. The
ore of aluminium is called bauxite, a brown, rusty coloured mineral containing
aluminium oxide or alumina (Al2O3). Bauxite is
usually found in areas of high rainfall and is the product of leaching
aluminium oxide from surface rock over millions of years. Alumina is
first chemically separated from bauxite ore by dissolving the bauxite
in sodium hydroxide solution, filtering off the unwanted coloured components
and then extracting the alumina by precipitation.
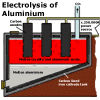 Aluminium
oxide, or alumina, is a very stable compound and requires lots of energy
to extract the aluminium metal. This energy is best supplied by passing
an electric current through a molten mixture of alumina and cryolite
(sodium aluminium fluoride) which melts and "dissolves" the
alumina at a lower temperature than would would be required otherwise.
The electric current is passed though the cell, or vessel holding the
molten mixture, from one electrode (the cathode) to another electrode
(the anode). Aluminium
oxide, or alumina, is a very stable compound and requires lots of energy
to extract the aluminium metal. This energy is best supplied by passing
an electric current through a molten mixture of alumina and cryolite
(sodium aluminium fluoride) which melts and "dissolves" the
alumina at a lower temperature than would would be required otherwise.
The electric current is passed though the cell, or vessel holding the
molten mixture, from one electrode (the cathode) to another electrode
(the anode).
 Aluminium
is produced at the cathode by positively charged aluminium ions accepting
free electrons to form aluminium atoms. At the anode, electrons are
effectively stripped of negatively charged oxide ions, which then combine
with carbon at the anode to produce carbon dioxide gas. Aluminium
is produced at the cathode by positively charged aluminium ions accepting
free electrons to form aluminium atoms. At the anode, electrons are
effectively stripped of negatively charged oxide ions, which then combine
with carbon at the anode to produce carbon dioxide gas.
The aluminium is then drained from the cell and the carbon dioxide
escapes to the atmosphere. This process consumes so much electricity,
that aluminium is sometimes referred to as "solid electricity"!
Electrolysis and other metals
Electrolysis is the most powerful way of releasing a metal from its
ore and can be used to obtain any metal from its salt. It is, however,
a very costly process and consumes vast quantities of electricity. Industrially,
only the most reactive metals are obtained in this way. Other metals
produced by electrolysis include, lithium, potassium, sodium, calcium
and magnesium.
|