|
Making molecules - the rules of
covalency
Molecules have formulas that are set by the capacity
of their atoms to form links or bonds with other atoms. The topic From
atoms to molecules has many examples of molecules and their formulas.
This topic investigates how these formulas are calculated from our knowledge
of atoms and their ability to form bonds.
 The ability of an atom to form bonds within a molecule is known
as covalency. Although covalency is very similar to the idea of electrovalency,
the two are often confused. Covalency is used for molecules and electrovalency
is used for ionic solids or salts. Electrovalency is discussed in the
topic Making
salts - the rules of electrovalency.
The ability of an atom to form bonds within a molecule is known
as covalency. Although covalency is very similar to the idea of electrovalency,
the two are often confused. Covalency is used for molecules and electrovalency
is used for ionic solids or salts. Electrovalency is discussed in the
topic Making
salts - the rules of electrovalency.
Covalency
The number of bonds an atom can form within a molecule
is known as its covalency. The simplest atom, hydrogen has the capacity
to form one bond; that is, it can attach itself to one other atom to
form a molecule. Oxygen atoms can form two bonds and nitrogen atoms
can form three within a molecule.
Here is a list of common atoms which form molecules with their covalency:
|
1
|
2
|
3
|
4
|
|
hydrogen
|
oxygen
|
nitrogen
|
carbon
|
|
fluorine
|
sulfur
|
phosphorus
|
silicon
|
|
chlorine
|
|
|
|
Matching covalencies
To calculate the formula for a molecule the covalencies of the atoms
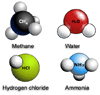
must be balanced. Take for instance the molecule methane (CH4)
made from carbon and hydrogen atoms. Carbon with covalency 4 is matched
by 4 hydrogen atoms each with covalency 1.
Hint:- It is most common for the atom with the highest covalency to
form the central atom in the molecule.
Ammonia is made of nitrogen and hydrogen. The central nitrogen atom
will combine with three single hydrogen atoms to form NH3.
Water is made of oxygen and hydrogen. The central oxygen atom will
combine with two single hydrogen atoms to form H2O.
Hydrogen chloride is made of chlorine and hydrogen. The chlorine atom
will combine with a single hydrogen atom to form HCl.
More combinations
Not all bonds between atoms in a molecule have to be single connections.
Many molecules have double and triple bonds between atoms. Double and
triple bonds in general are stronger than single bonds between the same
atoms. However double or triple bonds are usually much more reactive.
Carbon dioxide (CO2) is a molecule with multiable bonds.
Its central carbon atom, covalency 4 has two attached oxygen atoms,
each with covalency 2. The covalencies are balanced, ie. 4 = (2x2).
Try the quiz questions for more examples. Remember the idea is a simple
one, although the theory behind it is quite complex. In simple terms,
molecules match the covalency of the central atom with those of the
surrounding atoms, adding atoms until a balance of covalencies is achieved.
|