|
Changes in state - boiling, evaporation
and
condensation
 Boiling, evaporation and condensation are terms
that refer to the changes in state between liquids and gases:
Boiling, evaporation and condensation are terms
that refer to the changes in state between liquids and gases:
- Boiling occurs when a liquid is heated and turns to a gas.
- Evaporation occurs when a liquid changes to a gas at a temperature
below its normal boiling point.
- Condensation occurs when a gas is cooled and turns to a liquid.
Boiling - liquid to gas
As a liquid is heated, more and more particles will
evaporate off the liquid surface. It is when the pressure of these escaping
particles is equal to the external air pressure on the surface of the
liquid, that bubbles of gas start to form in the liquid.
Boiling occurs when enough particles in a liquid escape en masse from
the liquid to form bubbles of gas in the body of the liquid. These gas
bubbles then rise to the surface of the liquid and the liquid is said
to boil.
Boiling point
The boiling point (or condensation point) of a substance is defined
as the temperature at which the vapour pressure of the liquid is exactly
equal to the external pressure. Above the boiling point, the substance
exists as a gas and below, it exists predominately as a liquid.
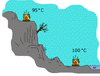 The
temperature at which a liquid boils is affected by atmospheric pressure.
The higher the altitude, the lower the temperature at which boiling
occurs (and the lower the liquid's boiling point). The
temperature at which a liquid boils is affected by atmospheric pressure.
The higher the altitude, the lower the temperature at which boiling
occurs (and the lower the liquid's boiling point).
When quoting a boiling point, scientists also quote the air pressure.
All standard boiling points are measured at sea level where the air
pressure is an average of 1 atmosphere or 101.3 kPa pressure.
Different liquids have different boiling points depending on the strength
of bonding between the particles and the mass of the particles. The
heavier the particles in the liquid, and the stronger the bonding, the
higher the boiling point will be.
Helium, the lightest and weakest bonded substance known, has a boiling
point very near absolute zero at -269°C. At the other end of the
scale, Tungsten, made of much heavier particles with very strong bonding,
boils at 5660°C.
 Boiling
point of pure and impure substances Boiling
point of pure and impure substances
For pure substances, the temperature at which boiling and condensing
occurs is quite sharp. For impure substances, boiling and condensing
occurs more gradually over a range of temperatures.
This is one way that chemists can measure the purity of a substance.
A pure substance will boil at a specific temperature, the more impure
a substance the more its boiling point will vary over a range of temperature.
Evaporation
When a liquid is heated, its particles eventually gain enough energy
to overcome the forces holding them loosely in place in the liquid and
become free, fast moving individual particles in the gas state.
Evaporation occurs when particles in a liquid pass directly into the
gas phase, at a temperature below the boiling point of the liquid. Evaporation
is dependent on individual particles gaining enough energy to escape
the surface of the liquid and becoming gas particles.
Condensation - gas to liquid
If a gas is cooled, its particles will eventually stop moving about
fast and form a liquid. This is called condensation and occurs at the
same temperature as boiling. The boiling point and condensation point
of a substance is the same temperature.
|