|
Atoms
Atoms are the basic building blocks of matter. Every thing is made
of atoms, rocks, living things, the Earth, the stars, all that we can
see. What are atoms made of?
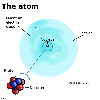 Subatomic
particles Subatomic
particles
The modern quantum atomic theory of the atom states
that all atoms have three fundamental parts (or subatomic particles):
protons, neutrons and electrons. All three can exist by themselves,
but it is when they combine to form atoms that they become truly spectacular
in their behavior.
The nucleus
The protons and neutrons are found tightly bound into a very small,
positively charged, central core called the nucleus. In the average
size atom, the nucleus takes up 100 000th of the diameter of the atom.
If the nucleus of an atom was the size of a marble, then the atom it
belonged to would be about the size of the a football ground. The nucleus
is so dense that if the marble in the middle of the ground was made
of only protons and neutrons it would weigh about 1 000 tonnes.
This extremely small, very dense centre of the atom has nearly all
of the atom's mass and is virtually indestructible in everyday chemical
and physical reactions. It is only in the stars, nuclear reactors, atomic
bombs and during radioactive decay that nuclei can be broken and reformed.
The electrons
The nucleus is surrounded by a "cloud" of very fast moving
electrons. In contrast to the nucleus, this vast empty space has virtually
no matter in it and hence no mass. The light weight electrons can move
very fast, some thousands of kilometres per hour, and act a bit like
the blades of a fan. When the fan is stopped, you can see the spaces
between the blades but, when it is full on, the blades and space blur
together and the blades effectively occupy all the available space.
The electrons are arranged in energy levels and orbitals, or regions
in space where electrons can be found. Scientists describe the arrangement
of electrons using mathematics and "quantum numbers" which
uniquely describe the shape of each orbital and the properties of each
electron, the modern quantum theory goes on to describe a strange subatomic
world of wave/particles and electromagnetic energy.
The atom
There are over 100 known types of atoms in our Universe, all made up
of the three subatomic particles. Each type of atom is identified primarily
by the number of protons it has in its nucleus; for example, the hydrogen
atom has 1 proton, while the uranium atom has 92 protons. The number
of protons an atom has is specified by its "Atomic Number",
and it is by using atomic numbers that scientists can arrange the atoms
into some order.
Each atom will also have just enough neutrons to hold all the protons
together in the nucleus. The presence of the neutrons allows two fundamental
forces, the weak and strong nuclear forces, to come into play and form
the tightly bound atomic nucleus.
The atom is made complete by adding the same number of electrons as
it has protons. This makes the whole atom electrically neutral. The
electrons are much more weakly bound to the atom and can move from one
atom to another or even be "rubbed off" an atom. Static electricity
is created by electrons rubbing off atoms and moving to other atoms.
It is these electrons which form the basis for chemical change and the
diversity of materials and substances we have in our modern world.
|