|
Acids and bases
Acids and bases play a central role in much of the chemistry of every
day life. Acids are found in many foods, like citrus fruits, soft drinks
and vinegar, while bases are found in cleaning agents and antacids.
Acids, bases, and water
The chemistry of acids and bases most commonly happens
in water.
The balance of acids and bases is crucial for our good health and for
our environment. If one dominates too much over the other, all sorts
of things go wrong and life on Earth would be in peril. Acid rain, fish
deaths from acid rain, dirty swimming pools, legionaire bacteria outbreaks
and indigestion are all examples of acids and bases being out of balance.
Acids are substances that when dissolved in water release hydrogen
ions, H+(aq).
Bases are substances that react with and neutralise acids, producing
water. When dissolved, bases release hydroxide ions, OH-(aq)
into solution. Water is the product of an acid and base reacting. Chemists
say that the acid and base cancel or neutralise each other, hence the
reaction is known as "neutralisation".
The simple equation for the neutralisation reaction between an acid
and a water soluble base is:
H+(aq) + OH-(aq)  H2O
H2O
Acids
Acids give foods a sour taste. Food acids are weak acids and are not
to be confused with the much stronger corrosive laboratory and industrial
acids like hydrochloric acid HCl, or sulfuric acid H2SO4,
which if ingested would be fatal.
 Acids
react with the more reactive metals to release hydrogen gas and can
change the colour of some chemical dyes. We can use this property to
test for the presence of an acid in solution. Acids
react with the more reactive metals to release hydrogen gas and can
change the colour of some chemical dyes. We can use this property to
test for the presence of an acid in solution.
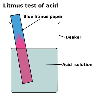
One such dye, litmus, turns from blue to red in the presence of an
acid and is the safest and simplest test for an acid. An acid is a substance
that turns blue litmus red.
Bases
Bases are not common in the biological world as the chemistry of life
has developed neutral or slightly acidic solutions to deal with most
processes. Sodium carbonate and washing soda are properly used household
bases. Bases can be either water soluble or insoluble. Insoluble bases
react with acids, directly dissolving in the acid as they react. Soluble
bases form hydroxide ions in solution, that then react with the acid
as described in the above section Acids, bases
and water.
Caution: Base solutions
are slippery or soapy to the touch. What is happening is the base is
reacting with the oils in your skin to form soap, and it is your own
oils that you are feeling as they turn into soap! Do not touch bases
or get them near your eyes.
 Some
common bases are sodium hydroxide NaOH, calcium hydroxide Ca(OH)2
and ammonia solution NH4OH. Sodium hydroxide is particularly
strong base and should not be handled at all. Some
common bases are sodium hydroxide NaOH, calcium hydroxide Ca(OH)2
and ammonia solution NH4OH. Sodium hydroxide is particularly
strong base and should not be handled at all.
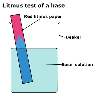
Bases are substances that neutralise acids and if dissolved in water,
turn red litmus blue.
Neutralisation
When an acid and a base are combined, water and a salt are formed. For
example, when hydrochloric acid is mixed with sodium hydroxide, water
and sodium chloride are formed.
The equation for this reaction is:
HCl + NaOH  H2O + NaCl
H2O + NaCl
Both hydrochloric acid and sodium hydroxide would dissolve you if you
fell into them, but you can go swimming in the product of their reaction,
salt water! Before you start making your own oceans from acids and bases,
remember; it is only when all of the acid has been neutralised by all
of the base that you will have neutral salt water. If one or the other
is in excess, then the solution will be salty, but it will also be either
acidic or basic as well.
Read the next topic, Measuring
acids and bases, for more on neutralisation and acid and base strength.
|